safety, driven by dedication, and delivering innovation.
How does a leak detector respond when hazardous inorganic solvents such as ammonia or hydrofluoric acid leak? This technical guide explains the scientific principles of ionic conductivity and dielectric constant, and presents a practical sensor selection guide based on the properties of hazardous chemicals.
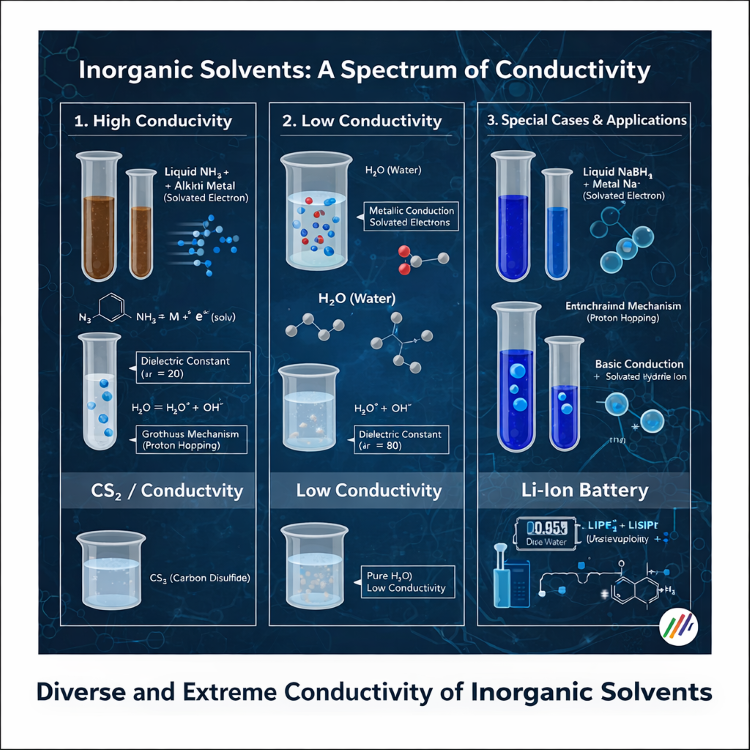
Hello from Yoohantech. In this article, we take a deeper technical look at the electrical conductivity of inorganic solvents, one of the core principles behind how a chemical leak detector works in industrial environments. Beyond simple water detection, we explain how a leak sensor responds to hazardous substances such as ammonia and hydrofluoric acid, and the science behind that response.
1. What Leak Detectors Sense: The Conductivity Spectrum of Solvents
Leak detectors identify spills by analyzing the electrical properties of a liquid. Inorganic solvents range from low-conductivity nonpolar substances to highly conductive polar liquids, and the appropriate sensor type depends on these characteristics.
2. Two Technical Principles Behind Leak Sensor Operation
The key to sensor response during a chemical leak is the generation and mobility of ions. Yoohantech sensors are designed to capture these subtle changes.
1. Ion Transfer Mechanism (Proton Jump) and Detection Speed
When a polar solvent leaks, it can self-ionize and carry charge. The leak detector reads this charge flow.
-
Ionization reaction: H2O + H2O ⇔ H3O+ + OH- (conductivity appears when leakage occurs)
-
Intrinsic conductivity and sensor sensitivity: Pure water has very low conductivity (0.055µS/cm), but the conductivity rises sharply when chemicals are mixed into it. A high-performance leak sensor can detect this subtle change.
-
Grotthuss mechanism: Because protons move rapidly through a hydrogen-bond network by “jumping,” known as the Grotthuss mechanism, a leak detector may respond even faster to acidic liquid leaks.
2. High Dielectric Constant and Sensor Response
Solvents such as water (εr ≈ 80) have a high dielectric constant, allowing ions to separate easily.
-
Ion dissociation: When a solvent separates ions, current flows between the electrodes of the leak detection sensor, triggering an alarm.
-
Stable detection: Solvation stabilizes ion movement, helping the sensor identify a liquid leak accurately without false operation.
3. Detection Mechanisms for Special Chemical Leaks
Some special liquids are difficult to detect with a standard leak detector, so understanding their unique behavior is essential.
1. Liquid Ammonia (NH3) Leak
When ammonia dissolves alkali metals, it forms solvated electrons, producing metallic-like conductivity. In such cases, a highly sensitive and specially engineered ammonia leak detector is required rather than a general-purpose sensor.
-
Reaction:
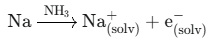
-
Result: Since charge is carried by free electrons rather than only ions, Yoohantech designs sensors with this distinctive property in mind.
2. Non-Conductive Solvents (Organic Solvents)
Nonpolar organic solvents such as carbon disulfide (CS2) behave almost like insulators. Therefore, accurate detection requires an electronic leak detector using a chemical-response method rather than simple conductivity-based sensing.
4. Conclusion: Choosing the Right Leak Detector for Safer Facilities
In chemistry and materials science, the conductivity characteristics of inorganic solvents form the foundation of leak detector development. Yoohantech analyzes each liquid’s conductivity behavior, including ion generation efficiency and electron transport, to provide a high-performance leak detection system optimized for water, acids, alkalis, and organic solvents.
To maintain safe operations, it is essential to install the right leak sensor for the properties of the target liquid.
![]()
🌌 Advanced Note: Sensor Response Mechanism During Ammonia (NH3) Leakage
Liquid ammonia (NH3) can cause severe damage when released as a hazardous chemical. In particular, when combined with alkali metals, the electrical signal that a leak detector must capture changes dramatically.
1. Leak Sensor Signal Changes by Concentration
The conductivity pattern detected by a leak detector changes depending on the concentration of the ammonia leak.
A. Low-Concentration Leak: Ionic Conduction
At the initial stage of leakage, a small amount of metal species may react and form ions.
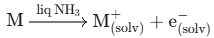
-
General detection: The leak sensor detects the physical movement of solvated ions (M+) and electrons, then activates an alarm.
-
Visual change: Solvated electrons appear blue, which can also provide a visual clue during inspection.
B. High-Concentration Leak: Metallic Conduction
As the concentration increases during a major leak, conductivity rises sharply and the leak detector sends an immediate danger signal.
-
Ultra-fast response: Because delocalized electrons move like those in a metal, the sensor’s conductivity reading can cross the alarm threshold almost instantly.
-
Importance of safety management: To prevent major accidents, facilities need a precision industrial leak detector capable of identifying these abrupt property changes.
Technical Conclusion
Understanding the conductivity mechanisms of chemical solvents is crucial for the development and proper selection of high-performance leak detectors. Based on this fundamental science, Yoohantech provides reliable safety solutions designed for real industrial environments.
Liquid Leak Detector: Technical Inquiry
We confirm applicability and recommend solutions based on your target liquid and site conditions.
Setting the trusted standard for liquid leak detection in industrial facilities.
I want to
Defining the Trusted Standard in Liquid Leak Detection for Industrial Safety.












