Why can organic solvents such as ethanol and hexane not be detected by ordinary sensors? This article explains the relationship between dielectric constant and conductivity, and provides a practical selection guide for choosing the right organic-solvent leak detector to help prevent chemical accidents.

Today’s topic for chemical plant and laboratory safety managers is 'the non-conductive nature of organic solvents and leak detectors'. Why are solvents such as ethanol, acetone, and hexane difficult to detect with ordinary water leak sensors? Let’s explore the scientific reason behind this and understand why choosing the right chemical leak detector is so important.
1. The Basics of Leak Detector Operation: The Principle of Conductivity
Most leak detectors work by using the ability of a liquid to conduct electricity. For a material to have conductivity, one of the following conditions is generally required:
-
Free Electrons: Electrons must be able to move freely, as they do in metals.
-
Mobile Ions: Ions in the liquid must be able to move and carry charge. This is the common principle used in standard acid/alkali leak detectors.
However, organic solvents do not sufficiently satisfy these conditions and therefore behave as non-conductive materials (insulators). For this reason, leaks involving organic solvents should not rely on conductivity-based sensing, but instead require an organic-solvent-specific leak detector.
2. Why Can’t Organic Solvents Be Detected by Ordinary Sensors?
The technical reasons why ordinary water leak sensors cannot detect organic solvents are as follows:
1. Low Autoionization
Most organic solvents are covalently bonded substances.
-
Water self-ionizes and can therefore conduct electricity, but organic solvents ionize only to an extremely small extent.
-
For example, ethanol has a very small ionization constant, around 10-20, which is far too low to generate enough current to activate a conductivity-based leak sensor.
2. Low Dielectric Constant
An important factor in designing a chemical leak detector is the dielectric constant.
-
If the dielectric constant is low, it cannot effectively weaken the attraction between ions.
-
According to Coulomb’s law (
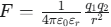 ), ions in low-dielectric organic solvents such as hexane and benzene do not separate easily and instead form ion pairs.
), ions in low-dielectric organic solvents such as hexane and benzene do not separate easily and instead form ion pairs. -
Result: Charge movement is blocked and conductivity remains low, so a low-cost conductivity-based leak sensor becomes ineffective.
3. The Right Leak Detection Solution
For facilities handling organic solvents, reliable safety requires installing a chemical-reaction-type organic solvent leak detector that does not depend on conductivity.
3. Special Case: Detection of Battery Electrolyte Leaks
Then how can organic electrolytes used in EV batteries and similar applications be detected?
💡 Point: Lithium-ion battery electrolytes contain a high concentration of electrolyte salt such as LiPF6 dissolved in organic solvents, which artificially increases conductivity. Because of this, leakage caused by battery damage can often be detected even by a general conductivity-based leak detector.
However, for pure organic solvent storage tanks and piping, it is essential to use an organic-solvent-specific sensor.
![]()
🔍 Advanced Guide: How to Choose a Leak Detector by Organic Solvent Type (Dielectric Constant Analysis)
The dielectric constant (εr) is a useful reference for determining whether a liquid may respond to a conductivity-based leak sensor or requires an organic-solvent-specific sensor.
1. Predicted Sensor Response by Major Solvent Type
The table below provides a practical reference for selecting the appropriate leak detector for different solvents.
| Solvent Type | Dielectric Constant (εr) | Leak Detector Selection Point |
|---|---|---|
| Water (H2O) | ≈ 80 | High conductivity, so a general water leak sensor can be used |
| Ethylene Carbonate | ≈ 90 | High polarity, so conductivity sensor response may be possible |
| Acetone | ≈ 21.5 | Low conductivity. Organic-solvent-specific sensor recommended |
| Ethanol | ≈ 25 | May become conductive when mixed with water, but pure ethanol requires a dedicated sensor |
| Dimethyl Carbonate | ≈ 3.1 | Almost no conductivity. An organic solvent leak detector is essential |
| Benzene | ≈ 2.3 | Completely non-polar. An organic-solvent-specific sensor must be used |
-
High εr: Ion separation is easier, so conductivity-based detection may be possible.
-
Low εr (most organic solvents): Strong attraction between ions makes the liquid behave more like an insulator. In such cases, ordinary electrode-rod type leak sensors cannot detect leaks reliably, increasing accident risk.
2. Key Technology for Preventing Chemical Leak Accidents
Although pure organic solvents have very low conductivity, industrial sites still need reliable ways to overcome this limitation and maintain safety.
1. Electrolyte Dissolution and Detection (Battery Process)
-
In battery manufacturing and similar processes, salts such as LiPF6 are added to the solvent.
-

-
In this case, the leak detector can trigger an alarm by detecting the flow of the generated ions.
2. Selecting the Right Leak Detector (Considering Dielectric Constant and Viscosity)
YOOHANTECH recommends the most suitable sensor according to the characteristics of the liquid.
-
Conductive Liquids: Acids, alkalis, and water are suitable for fast-response conductivity sensors.
-
Non-Conductive Organic Solvents: Benzene, toluene, and similar liquids have low conductivity, so they require an organic-solvent-specific leak detector based on chemical reaction principles.
| Solvent Characteristic | Dielectric Constant (εr) | Detection Difficulty | Recommended Leak Detector Type |
|---|---|---|---|
| High Dielectric | High | Easy | Point Sensor for Inorganic / Conductive Liquids |
| Low Dielectric | Low | Difficult | Point Sensor for Organic Solvents |
Liquid Leak Detector: Technical Inquiry
We confirm applicability and recommend solutions based on your target liquid and site conditions.
Setting the trusted standard for liquid leak detection in industrial facilities.